
Chemistry, 05.05.2020 01:16 jetthehawk9861
A scientist adds 46.3 J of heat to a 13.4g sample of an unknown substance. the temperature increases by 1.40 celsius. What is the unknown substance?
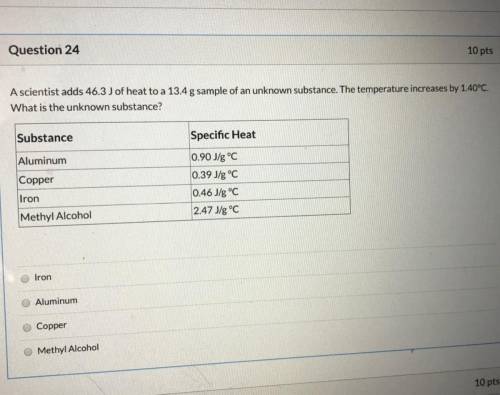

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:00, Ashleyvasquez2261
Type the letter that represents the correct location for each particle type below.
Answers: 1

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1
You know the right answer?
A scientist adds 46.3 J of heat to a 13.4g sample of an unknown substance. the temperature increases...
Questions in other subjects:



Mathematics, 03.10.2019 08:10


History, 03.10.2019 08:10

Geography, 03.10.2019 08:10






