27.0°C and 1.00 atm. What will the volume be at

Chemistry, 05.05.2020 01:01 coxtinam16
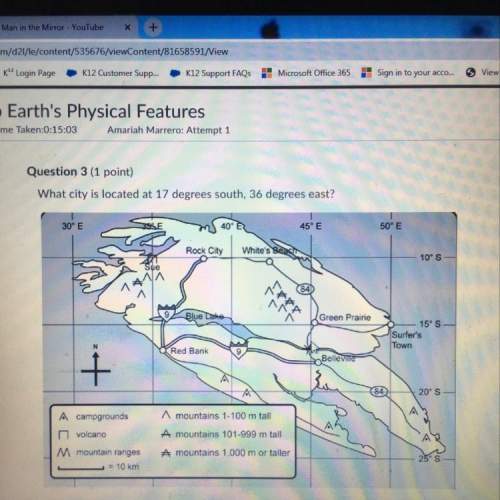
A sample of nitrogen gas occupies 1.55 L at
27.0°C and 1.00 atm. What will the volume be at
−100.0°C and the same pressure?
(2 Points)
0.894 L
0.998 L
0.58 L

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, rntaran2002
What is the ph of a 0.0010 m hno3? 1.0 3.0 4.0 5.0
Answers: 2

Chemistry, 22.06.2019 12:40, carebear60
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1
You know the right answer?
A sample of nitrogen gas occupies 1.55 L at
27.0°C and 1.00 atm. What will the volume be at
27.0°C and 1.00 atm. What will the volume be at
Questions in other subjects:





Mathematics, 23.05.2020 20:59

History, 23.05.2020 20:59

English, 23.05.2020 20:59


History, 23.05.2020 20:59