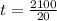Chemistry, 05.05.2020 06:35 genyjoannerubiera
Zinc is to be electroplated onto both sides of an iron sheet that is 20 cm2 as a galvanized sacrificial anode. It is desired to electroplate the zinc to a thickness of 0.025 mm. It is found that a current of 20 A produces a zinc coating of sufficient quality for galvanized iron. Determine the time required to produce the desired coating, assuming 100 % efficiency.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2


Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2
You know the right answer?
Zinc is to be electroplated onto both sides of an iron sheet that is 20 cm2 as a galvanized sacrific...
Questions in other subjects:




Chemistry, 08.02.2021 22:40

English, 08.02.2021 22:40




Mathematics, 08.02.2021 22:40

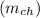 is given as :
is given as :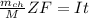
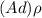 ; replacing that into above equation; we have:
; replacing that into above equation; we have: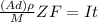 ---- equation (1)
---- equation (1)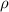 = density
= density ![[ \frac{20 cm^2 *0.0025 cm*7.13g/cm^3}{65.38g/mol}*2 \frac{moles\ of \ electrons}{mole \ of \ Zn} * 9.65*10^4 \frac{C}{mole \ of \ electrons } ] = (20 A) t](/tpl/images/0634/2762/37add.png)