
Chemistry, 05.05.2020 16:30 hhhhhh8897
21-B. Mn was used as an internal standard for measuring Fe by atomic absorption. A standard mixture containing 2.00 mg Mn/mL and 2.50 mg Fe/mL gave a quotient (Fe signal/Mn signal) 5 1.05/1.00. A mixture with a volume of 6.00 mL was prepared by mixing 5.00 mL of unknown Fe solution with 1.00 mL containing 13.5 mg Mn/mL. The absorbance of this mixture at the Mn wave- length was 0.128, and the absorbance at the Fe wavelength was 0.185. Find the molarity of the unknown Fe solution.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, Aminton737
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 12:20, tenleywood
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2

Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
You know the right answer?
21-B. Mn was used as an internal standard for measuring Fe by atomic absorption. A standard mixture...
Questions in other subjects:





Biology, 30.10.2020 01:50

Mathematics, 30.10.2020 01:50

Mathematics, 30.10.2020 01:50

Mathematics, 30.10.2020 01:50

Mathematics, 30.10.2020 01:50

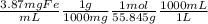 = 0.0693M Fe
= 0.0693M Fe

