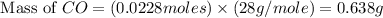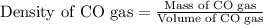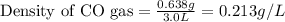Chemistry, 05.05.2020 16:12 batmanmarie2004
A quantity of CO gas occupies a volume of 0.64 L at 0.90 atm and 307 K . The pressure of the gas is lowered and its temperature is raised until its volume is 3.0 L .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, petriajack8375
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 22.06.2019 14:00, njones58emailtjcedu
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 20:20, Matseleng3775
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1
You know the right answer?
A quantity of CO gas occupies a volume of 0.64 L at 0.90 atm and 307 K . The pressure of the gas is...
Questions in other subjects:

Physics, 03.11.2020 01:00

Mathematics, 03.11.2020 01:00


Advanced Placement (AP), 03.11.2020 01:00

Mathematics, 03.11.2020 01:00

Social Studies, 03.11.2020 01:00



Chemistry, 03.11.2020 01:00

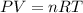
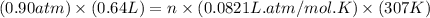
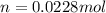
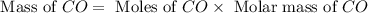
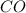 = 28g/mole
= 28g/mole