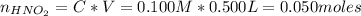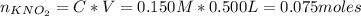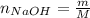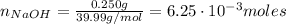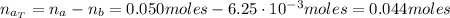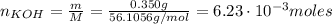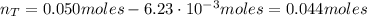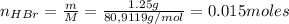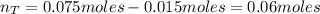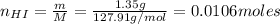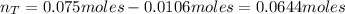A 500.0-mL buffer solution is 0.100 M in HNO2 and 0.150 M in KNO2. Part A Determine whether or not 250 mgNaOH would exceed the capacity of the buffer to neutralize it. Determine whether or not 250 would exceed the capacity of the buffer to neutralize it. yes no Request Answer Part B Determine whether or not 350 mgKOH would exceed the capacity of the buffer to neutralize it. Determine whether or not 350 would exceed the capacity of the buffer to neutralize it. yes no Request Answer Part C Determine whether or not 1.25 gHBr would exceed the capacity of the buffer to neutralize it. Determine whether or not 1.25 would exceed the capacity of the buffer to neutralize it. yes no Request Answer Part D Determine whether or not 1.35 gHI would exceed the capacity of the buffer to neutralize it. Determine whether or not 1.35 would exceed the capacity of the buffer to neutralize it. yes no

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 05:00, adjjones2011
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
A 500.0-mL buffer solution is 0.100 M in HNO2 and 0.150 M in KNO2. Part A Determine whether or not 2...
Questions in other subjects:

English, 24.07.2021 02:40








Physics, 24.07.2021 02:50