Chemistry, 05.05.2020 17:34 shamim5364
Iron (III) oxide and hydrogen react to form iron and water, like this: Fe 03(s)+3H9)2Fe(s)+3HO) At a certain temperature, a chemist finds that a 8.9 L reaction vessel containing a mixture of iron(III) oxide, hydrogen, Iron, and water at equilbrium has the following composition compound amount Fe 3.95 g H 4.77 g Fe 4.38 g H2 2.00 g Calculate the value of the equilibrium constant Kc for this reaction. Round your answer to 2 significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, mv603177
Rocks, as they are compressed, begin forming mountains above the earth's surface when two continental plates converge. the continental crust increases in depth as the mountains grow above. the himalayan mountains formed at a convergent plate boundary in this manner. the rocks are smashed together causing them to due to the intense heat and pressure from the colliding plates and eventually forming rock. a) melt; igneous b) layer; sedimentary c) recrystallize; metamorphic d) melt into the earth's interior; metamorphic
Answers: 1

Chemistry, 22.06.2019 14:40, neonbluefaith
Which statement best describes the function of enzymes?
Answers: 1

Chemistry, 22.06.2019 16:50, TrueKing184
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 22.06.2019 19:00, hmontalvo22
How many moles are contained in 5.6 l of h2 at stp
Answers: 3
You know the right answer?
Iron (III) oxide and hydrogen react to form iron and water, like this: Fe 03(s)+3H9)2Fe(s)+3HO) At a...
Questions in other subjects:


Social Studies, 27.04.2021 18:00

Chemistry, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00

English, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00


Biology, 27.04.2021 18:00

History, 27.04.2021 18:00

Mathematics, 27.04.2021 18:00

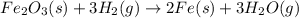
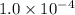
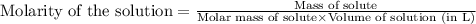
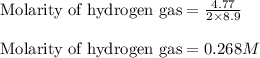
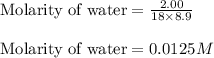
![K_{eq}=\frac{[H_2O]^3}{[H_2]^3}](/tpl/images/0640/8214/066de.png)