Chemistry, 05.05.2020 16:57 babygirl6571
The combustion of 1.760 g of propanol (C3H7OH) increases the temperature of a bomb calorimeter from 298.00K to 302.15K. The heat capacity of the bomb calorimeter is 14.24 kJ/K. Determine ΔH for the combustion of propanol to carbon dioxide gas and liquid water in kJ/mol.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, artemiscrock77041
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1


You know the right answer?
The combustion of 1.760 g of propanol (C3H7OH) increases the temperature of a bomb calorimeter from...
Questions in other subjects:

History, 04.08.2019 04:00


History, 04.08.2019 04:00



English, 04.08.2019 04:00


Mathematics, 04.08.2019 04:00


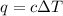
 = change in temperature =
= change in temperature = 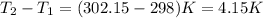
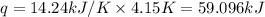
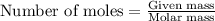
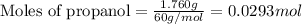
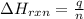
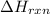 = enthalpy change of the reaction
= enthalpy change of the reaction