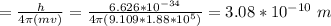Chemistry, 06.05.2020 01:59 silvajimena74
An electron travelling at 3.7 x 105 m/s has an uncertainty in its velocity of 1.88 x 105 m/s. What is the uncertainty in its position (in pm)? (Just put the number, don't use scientific notation and decimal point in the final answer) Avogadro's number = 6.022 x 1023, h = 6.626 x 10-34 J. s, c = 3.00 x 108 m/s, me= 9.109 x 10-31 kg

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, badgirl2005
This is a mixture that has the same composition throughout.
Answers: 1


Chemistry, 22.06.2019 22:30, pookie879
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

Chemistry, 23.06.2019 00:30, evelynalper08
Which radioisotope is used to date fossils? a. oxygen-16 b. carbon-14 c. uranium-238 d. carbon-12
Answers: 2
You know the right answer?
An electron travelling at 3.7 x 105 m/s has an uncertainty in its velocity of 1.88 x 105 m/s. What i...
Questions in other subjects:


Biology, 05.02.2022 08:40

History, 05.02.2022 08:40




Mathematics, 05.02.2022 08:40

English, 05.02.2022 08:40

Advanced Placement (AP), 05.02.2022 08:40

Mathematics, 05.02.2022 08:50