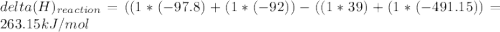Chemistry, 06.05.2020 06:36 jetblackcap
Hydroxyl radicals react with and eliminate many atmospheric pollutants. However, the hydroxyl radical does not clean up everything. For example, chlorofluorocarbons - which destroy stratospheric ozone - are not attacked by the hydroxyl radical. Consider the hypothetical reaction by which the hydroxyl radical might react with a chlorofluorocarbon: OH(g)+CF2Cl2(g)-->HOF(g)+CFCl2(g )Use bond energies to explain why this reaction is improbable. Calculate \Delta Hrxn of this reaction.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, bigwaYne
Imagine that twenty i. u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

You know the right answer?
Hydroxyl radicals react with and eliminate many atmospheric pollutants. However, the hydroxyl radica...
Questions in other subjects:

Mathematics, 28.01.2021 19:00

History, 28.01.2021 19:00

Spanish, 28.01.2021 19:00


History, 28.01.2021 19:00


Mathematics, 28.01.2021 19:00