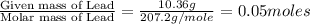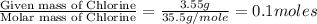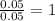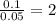Chemistry, 06.05.2020 07:29 jalexyinez
In an experiment similar to the zinc chloride experiment, a student placed a piece of lead in hydrochloric acid. Hydrogen gas was given off, and then the liquid was boiled off. The remaining solid, lead chloride was massed. Use the data below to determine the empirical formula of lead chloride.
mass of beaker 204.35 g
mass of lead and beaker before reaction 214.71 g
mass of lead chloride and beaker after reaction 218.26 g

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:40, Calumworthy6046
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 06:30, khalaflaf2684
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 22:20, icantspeakengles
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

Chemistry, 23.06.2019 00:20, destromero
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
In an experiment similar to the zinc chloride experiment, a student placed a piece of lead in hydroc...
Questions in other subjects:

Mathematics, 20.02.2021 22:10


Mathematics, 20.02.2021 22:10

Mathematics, 20.02.2021 22:10

Mathematics, 20.02.2021 22:10




Mathematics, 20.02.2021 22:10

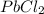
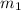 = 204.35 g
= 204.35 g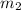 = 214.71 g
= 214.71 g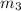 = 218.26 g
= 218.26 g = [214.71 - 204.35] = 10.36 g
= [214.71 - 204.35] = 10.36 g = [218.26 - 214.71] = 3.55 g
= [218.26 - 214.71] = 3.55 g