
Chemistry, 06.05.2020 06:58 LarryJoeseph
One type of breathalyzer employs a fuel cell to measure the quantity of alcohol in the breath. When a suspect blows into the breathalyzer, ethyl alcohol is oxidized to acetic acid at the anode:
CH3CH2OH(g)+4OH−(aq)→HC2H3O2(g)+3H2 O(l)+4e−
At the cathode, oxygen is reduced:
O2(g)+2H2O(l)+4e−→4OH−(aq)
The overall reaction is the oxidation of ethyl alcohol to acetic acid and water. When a suspected drunk driver blows 186 mL of his breath through this breathalyzer, the breathalyzer produces an average of 320 mA of current for 10 s.
Required:
Assuming a pressure of 1.0 atm and a temperature of 26C, what percent (by volume) of the driver's breath is ethanol?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, livigrace9004
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 07:30, bbyniah123
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
One type of breathalyzer employs a fuel cell to measure the quantity of alcohol in the breath. When...
Questions in other subjects:

Mathematics, 18.03.2021 02:50

Arts, 18.03.2021 02:50

Mathematics, 18.03.2021 02:50

Mathematics, 18.03.2021 02:50


History, 18.03.2021 02:50

Mathematics, 18.03.2021 02:50

Advanced Placement (AP), 18.03.2021 02:50




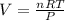

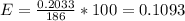 %
%

