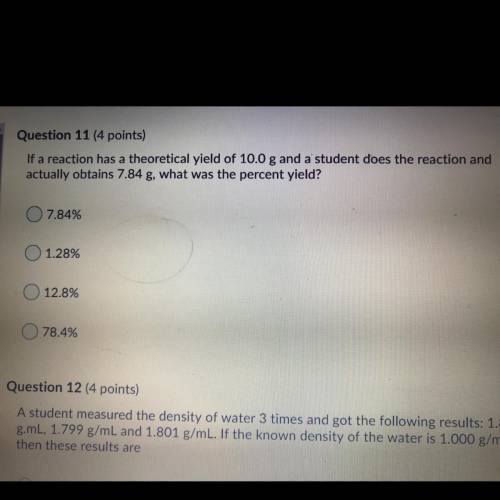
If a reaction has a theoretical yield of 10.0 g and a student does the reaction and
actually o...

Chemistry, 27.04.2020 01:39 marshallmattah
If a reaction has a theoretical yield of 10.0 g and a student does the reaction and
actually obtains 7.84 g, what was the percent yield?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3



Chemistry, 23.06.2019 08:00, codybrocs9624
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 2
You know the right answer?
Questions in other subjects:

English, 09.11.2020 22:10


History, 09.11.2020 22:10

Social Studies, 09.11.2020 22:10

Mathematics, 09.11.2020 22:10



Mathematics, 09.11.2020 22:10


Mathematics, 09.11.2020 22:10



