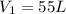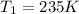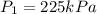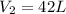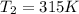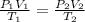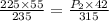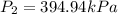Chemistry, 26.04.2020 03:48 zirconium16
A 55.0 L sample of gas at 235 K exerts a pressure of 225 KPa. If its volume is decreased to 42.0 L and the temperature rises to 315 K, what is its new pressure?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3


Chemistry, 22.06.2019 14:30, isaiahrodriguezsm17
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

You know the right answer?
A 55.0 L sample of gas at 235 K exerts a pressure of 225 KPa. If its volume is decreased to 42.0 L a...
Questions in other subjects:



Biology, 01.10.2019 13:30

Computers and Technology, 01.10.2019 13:30

English, 01.10.2019 13:30

Social Studies, 01.10.2019 13:30

History, 01.10.2019 13:30

World Languages, 01.10.2019 13:30