
Chemistry, 25.04.2020 23:13 samantha9430
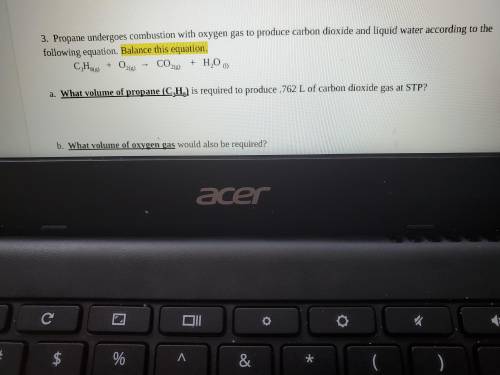
Propane undergoes combustion with oxygen gas to produce carbon dioxide and liquid water according to the following equation. Balance this equation.
C3H8(g) + O2(g) → CO2(g) + H2O (l)
a. What volume of propane (C3H8) is required to produce .762 L of carbon dioxide gas at STP?
b. What volume of oxygen gas would also be required?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, kayleg907436
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 05:30, alexusnicole817
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 09:00, boxergirl2062
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3
You know the right answer?
Propane undergoes combustion with oxygen gas to produce carbon dioxide and liquid water according to...
Questions in other subjects:


Physics, 22.05.2020 00:12





Mathematics, 22.05.2020 00:12


Mathematics, 22.05.2020 00:12

English, 22.05.2020 00:12



