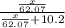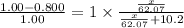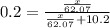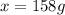Chemistry, 25.04.2020 04:06 amortegaa805
What mass of ethylene glycol, when mixed with 183 g H2O, will reduce the equilibrium vapor pressure of H2O from 1.00 atm to 0.800 atm at 100 °C? The molar masses of water and ethylene glycol are 18.02 g/mol and 62.07 g/mol, respectively. Assume ideal behavior for the solution.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, baileysosmart
The diagram shows the positions of the sun, moon and earth during spring tides, when the high tides are at their highest and low tides at their lowest. what is it about these positions that causes these high and low tides?
Answers: 3

Chemistry, 22.06.2019 01:30, elizediax8683
(apex) when a cup of water is dropped, as the cup falls, the water in the cup falls out true or false?
Answers: 1

Chemistry, 22.06.2019 11:50, hamidaakter936848
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2
You know the right answer?
What mass of ethylene glycol, when mixed with 183 g H2O, will reduce the equilibrium vapor pressure...
Questions in other subjects:

Physics, 02.03.2021 09:00

Mathematics, 02.03.2021 09:00

Mathematics, 02.03.2021 09:00

Mathematics, 02.03.2021 09:00

English, 02.03.2021 09:00

Mathematics, 02.03.2021 09:00


Social Studies, 02.03.2021 09:00

History, 02.03.2021 09:00

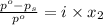
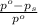 = relative lowering in vapor pressure
= relative lowering in vapor pressure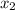 = mole fraction of solute =
= mole fraction of solute =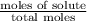
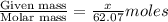
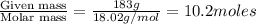
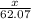 + 10.2
+ 10.2