
Chemistry, 25.04.2020 03:04 guapotaco8102
Consider two equal-volume flasks of gas at the same temperature and pressure. One gas, oxygen, has a molecular mass of 32. The other gas, nitrogen, has a molecular mass of 28. What is the ratio of the number of oxygen molecules to the number of nitrogen molecules in these flasks

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, fordkenae
24 points and brainliest to anyone who can answer under 10 minutes with best ! the table below shows the role of different substances during photosynthesis. substance role during photosynthesis glucose stores chemical energy water combines with glucose to form carbon dioxide chlorophyll traps sunlight which of the following statements would correct one of the roles listed in the table? glucose combines with carbon to form water. chlorophyll reacts with light to produce carbon dioxide. water combines with carbon dioxide during photosynthesis. chlorophyll stores chemical energy needed for photosynthesis.
Answers: 1

Chemistry, 22.06.2019 03:30, electrofy456
What diagram shows the ionic compound of magnesium oxide
Answers: 2


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
You know the right answer?
Consider two equal-volume flasks of gas at the same temperature and pressure. One gas, oxygen, has a...
Questions in other subjects:


English, 28.07.2019 18:30

History, 28.07.2019 18:30

Social Studies, 28.07.2019 18:30


Mathematics, 28.07.2019 18:30

Mathematics, 28.07.2019 18:30



Social Studies, 28.07.2019 18:30

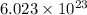 of particles.
of particles.

