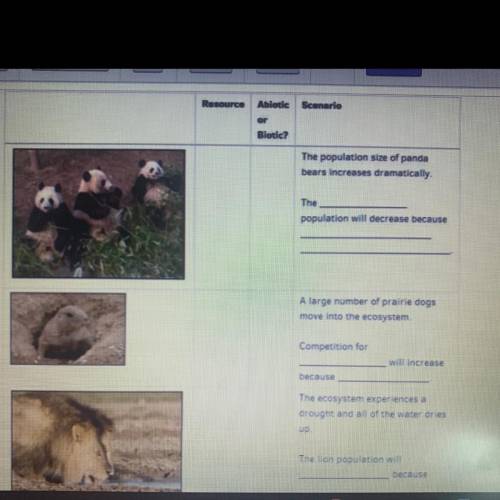
I need some help
can y’all help me
s ok
...

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 20.06.2019 18:04, brendanhein1
1. according to the article, the chinese government has tried to control its people in all of the following ways except: question 1 options: create and spread propaganda in the media censorship of the media allowing people to create their own social media deleting people's social media posts without their permission question 2 (1 point) 2. when a government has total control over its citizens, including the media, it is a of government question 2 options: constitutional monarchy parliamentary democracy autocracy presidential democracy question 3 (1 point) according to the article, "social credit" is question 3 options: an experiment to people talk on-line a farming experiment that hurt people under mao ze dong using someone else's data from the internet, to give or take away things such as the right to buy an airline ticket a credit you get in china for having friends in real life, and not just on social media question 4 (1 point) china wants to take control over social media in which other region? question 4 options: taiwan chile japan hong kong
Answers: 1

Chemistry, 21.06.2019 20:30, mvtthewisdead
If 10.g of agno3 is available, what volume of 0.25 m agno3 can be prepared
Answers: 1

Chemistry, 21.06.2019 23:30, 23gordns
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉mo re accurate estimates can be made with the van der waals equationí‘ťí‘ť=í‘›í‘›í‘›í‘›í‘›í‘›í‘ ‰í‘‰â’푛푛푟푟â’푞푞푛푛2í‘ ‰í‘‰2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 18:10, NEONREDBLADE
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3
You know the right answer?
Questions in other subjects:

Law, 03.12.2020 03:20

German, 03.12.2020 03:20



Mathematics, 03.12.2020 03:20

Mathematics, 03.12.2020 03:20

Mathematics, 03.12.2020 03:20



Engineering, 03.12.2020 03:20