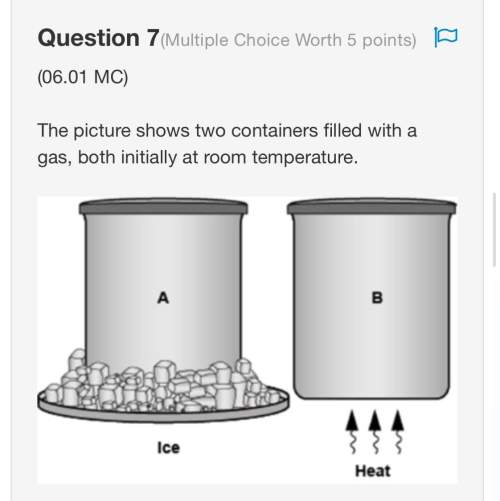
A sample of gas in a syringe has a volume of 9.66 mL at a pressure of
64.4 kPa. The plunger is...

Chemistry, 23.04.2020 10:46 aomoloju4202
A sample of gas in a syringe has a volume of 9.66 mL at a pressure of
64.4 kPa. The plunger is depressed until the pressure is 94.6 kPa. What
is the new volume, assuming constant temperature?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, taylorpayne525p8qxky
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2


Chemistry, 22.06.2019 20:00, bettybales1986
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 22.06.2019 20:30, sydneip6174
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
Questions in other subjects:

Mathematics, 26.09.2021 22:30



Mathematics, 26.09.2021 22:30


Mathematics, 26.09.2021 22:30

Mathematics, 26.09.2021 22:30

Mathematics, 26.09.2021 22:30


Health, 26.09.2021 22:30