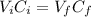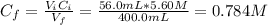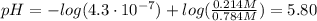Chemistry, 22.04.2020 22:28 ayowazzzgood
To prepare a buffer you weigh out 7.20 grams of NaHCO3 and place it into a 400.00 mL volumetric flask. To this flask you add 56.0 mL of 5.60 M H2CO3 and then fill it about halfway with distilled water, swirling to dissolve the contents. Finally, the flask is filled the rest of the way to the mark with distilled water. What is the pH of the buffer that you have created?Acid KaH2CO3 4.3 X 10⁻⁷ HCN 4.9 X 10⁻¹⁰HNO2 4.6 X 10⁻⁴C6H5COOH 6.5 X 10⁻⁵

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 15:30, officialrogerfp3gf2s
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1

Chemistry, 22.06.2019 05:00, YoEsMyles3115
0.2348 grams of pbcl2 used to form 44.0 ml of solution.
Answers: 1

Chemistry, 22.06.2019 18:00, sandeebassett3
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
You know the right answer?
To prepare a buffer you weigh out 7.20 grams of NaHCO3 and place it into a 400.00 mL volumetric flas...
Questions in other subjects:




Biology, 25.02.2020 19:59




Mathematics, 25.02.2020 19:59

Mathematics, 25.02.2020 19:59

History, 25.02.2020 19:59

![pH = pKa + log(\frac{[NaHCO_{3}]}{[H_{2}CO_{3}]})](/tpl/images/0619/4920/b4240.png) (1)
(1)![[NaHCO_{3}] = \frac{mol}{V} = \frac{m}{M*V}](/tpl/images/0619/4920/e7247.png)
![[NaHCO_{3}] = \frac{7.20 g}{84.007 g/mol*400.0 \cdot 10^{-3} L} = 0.214 M](/tpl/images/0619/4920/c7722.png)