
Chemistry, 22.04.2020 19:49 Xavierayala2003
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mole of CO2(g) that reacts. Write a balanced thermochemical equation for the reaction with an energy term in kJ as part of the equation. Note that the answer box for the energy term is case sensitive. Use the SMALLEST INTEGER coefficients possible and put the energy term in the last box on the appropriate side of the equation. If a box is not needed, leave it blank. 2CO2 + 5H2 + C2H2 + 4H2O + -46.6kJ

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 06:30, Liapis
An engineer decides to use a slightly weaker material rather than a stronger material, since she knows that the stronger material can break suddenly. this is an example of what? a choosing a material that will show warning before it fails b using composite materials that combine strength c using a material for multiple applications d using design techniques that increase efficiency and reduce cost
Answers: 3
You know the right answer?
When CO2(g) reacts with H2(g) to form C2H2(g) and H2O(g), 23.3 kJ of energy are absorbed for each mo...
Questions in other subjects:




English, 05.05.2020 16:29

History, 05.05.2020 16:29



Mathematics, 05.05.2020 16:29

Social Studies, 05.05.2020 16:29

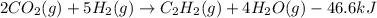
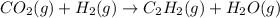
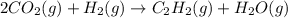
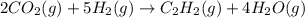
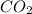 react. So, energy absorbed during the reaction is
react. So, energy absorbed during the reaction is 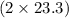 kJ or 46.6 kJ
kJ or 46.6 kJ


