
Chemistry, 22.04.2020 04:55 klanderos890
The reaction: 2 ClO2 (aq) + 2OH- (aq)→ ClO3- (aq) + ClO2- + H2O (l) was studied with the following results: Experiment [ClO2] (M) [OH-] (M) Initial Rate (M/s) 1 0.060 0.030 0.0248 2 0.020 0.030 0.00276 3 0.020 0.090 0.00828 a. Determine the rate law for the reaction. b. Calculate the value of the rate constant with the proper units. c. Calculate the rate when [ClO2] = 0.100 M and [OH-] = 0.050 M.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:00, aleilyg2005
If two objects at different te, peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 15:00, Zagorodniypolina5
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
The reaction: 2 ClO2 (aq) + 2OH- (aq)→ ClO3- (aq) + ClO2- + H2O (l) was studied with the following r...
Questions in other subjects:



Mathematics, 24.06.2021 05:30

Mathematics, 24.06.2021 05:30

Spanish, 24.06.2021 05:30

Mathematics, 24.06.2021 05:40

Mathematics, 24.06.2021 05:40

Mathematics, 24.06.2021 05:40

History, 24.06.2021 05:40

![\text v_o &= \text k [\text A]^x [\text B]^y](/tpl/images/0617/7012/bcd29.png)
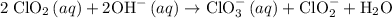
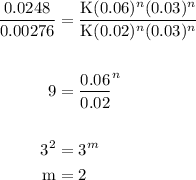
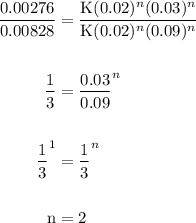
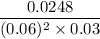 = 229.63 m⁻²s⁻¹
= 229.63 m⁻²s⁻¹



