Answer : The number of molecules of nitrogen, oxygen, carbon dioxide and argon is,  ,
, 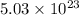 ,
,  and
and 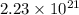 respectively.
respectively.
Explanation :
First we have to calculate the total moles of mixture of gas by using ideal gas equation.
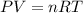
where,
P = Pressure of mixture of gas = 1.59 atm
V = Volume of mixture of gas = 6.51 L
n = number of moles mixture of gas = ?
R = Gas constant = 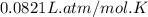
T = Temperature of mixture of gas = 
Putting values in above equation, we get:

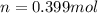
Now we have to calculate the moles of nitrogen, oxygen, carbon dioxide and argon.
Moles of nitrogen = 
Moles of oxygen = 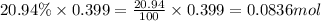
Moles of carbon dioxide = 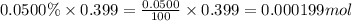
Moles of argon = 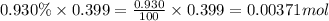
Now we have to calculate the number of molecules of nitrogen, oxygen, carbon dioxide and argon.
As, 1 mole of nitrogen contains 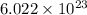 number of molecules of nitrogen.
number of molecules of nitrogen.
So, 0.312 mole of nitrogen contains 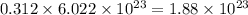 number of molecules of nitrogen.
number of molecules of nitrogen.
and,
As, 1 mole of oxygen contains 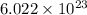 number of molecules of oxygen.
number of molecules of oxygen.
So, 0.0836 mole of oxygen contains 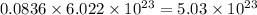 number of molecules of oxygen.
number of molecules of oxygen.
and,
As, 1 mole of carbon dioxide contains 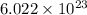 number of molecules of carbon dioxide.
number of molecules of carbon dioxide.
So, 0.000199 mole of carbon dioxide contains  number of molecules of carbon dioxide.
number of molecules of carbon dioxide.
and,
As, 1 mole of argon contains 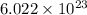 number of molecules of argon.
number of molecules of argon.
So, 0.00371 mole of argon contains 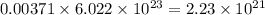 number of molecules of argon.
number of molecules of argon.
Therefore, the number of molecules of nitrogen, oxygen, carbon dioxide and argon is,  ,
, 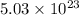 ,
,  and
and 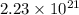 respectively.
respectively.


















 ,
, 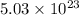 ,
,  and
and 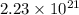 respectively.
respectively.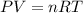
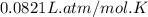


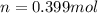

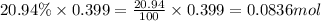
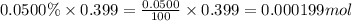
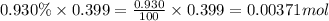
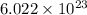 number of molecules of nitrogen.
number of molecules of nitrogen.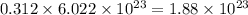 number of molecules of nitrogen.
number of molecules of nitrogen.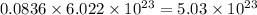 number of molecules of oxygen.
number of molecules of oxygen. number of molecules of carbon dioxide.
number of molecules of carbon dioxide.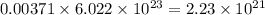 number of molecules of argon.
number of molecules of argon.

