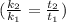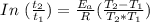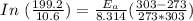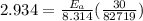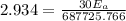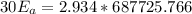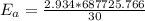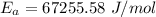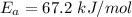Chemistry, 22.04.2020 03:28 wallacelizzy1
A food substance kept at 0°C becomes rotten (as determined by a good quantitative test) in 8.3 days. The same food rots in 10.6 hours at 30°C. Assuming the kinetics of the microorganisms enzymatic action is responsible for the rate of decay, what is the activation energy for the decomposition process? Hint: Rate varies INVERSELY with time; a faster rate produces a shorter decomposition time. 1.67.2 kJ/mol 2.2.34 kJ/mol 3.23.4 kJ/mol 4.0.45 kJ/mol

Answers: 3


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 16:30, joshua1255
Find the number of moles of argon in 364g of argon.
Answers: 2
You know the right answer?
A food substance kept at 0°C becomes rotten (as determined by a good quantitative test) in 8.3 days...
Questions in other subjects:

Mathematics, 05.05.2020 09:54




Mathematics, 05.05.2020 09:54


History, 05.05.2020 09:54

Mathematics, 05.05.2020 09:54

English, 05.05.2020 09:54

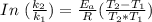
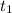 = 8.3 days = (8.3 × 24 ) hours = 199.2 hours
= 8.3 days = (8.3 × 24 ) hours = 199.2 hours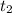 = 10.6 hours
= 10.6 hours 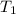 = 0° C = (0+273 )K = 273 K
= 0° C = (0+273 )K = 273 K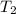 = 30° C = (30+ 273) = 303 K
= 30° C = (30+ 273) = 303 K