
Chemistry, 22.04.2020 01:27 Queenvalentin
A 25.888 g sample of aqueous waste leaving a fertilizer manufacturer contains ammonia. The sample is diluted with 73.464 g of water. A 10.762 g aliquot of this solution is then titrated with 0.1039 M HCl . It required 31.89 mL of the HCl solution to reach the methyl red endpoint. Calculate the weight percent NH3 in the aqueous waste.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, kingsqueen883
Consider the two electron arrangements for neutral atoms a and b. are atoms a and b the same element? a - 1s2, 2s2, 2p6, 3s1 b - 1s2, 2s2, 2p6, 5s1
Answers: 3



Chemistry, 22.06.2019 22:10, steven0448
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
You know the right answer?
A 25.888 g sample of aqueous waste leaving a fertilizer manufacturer contains ammonia. The sample is...
Questions in other subjects:

Mathematics, 28.09.2019 17:30


Mathematics, 28.09.2019 17:30


History, 28.09.2019 17:30

Mathematics, 28.09.2019 17:30






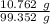
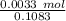
 %
%

