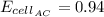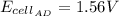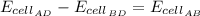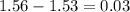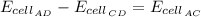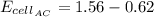Analyze and solve this partially completed galvanic cell puzzle. There are 4 electrodes each identified by a letter of the alphabet, A through D. The values in the partially completed grid are measured cell potentials for a cell consisting of electrode #1 and electrode #2. You may assume that each galvanic cell was properly constructed with the appropriate metals and solutions and that all the measured values in the grid are accurate.
electrode #1 ?
C
B
D
A
electrode #2?
Ecell(volts)
Ecell(volts)
Ecell(volts)
Ecell(volts)
C
0
0.91
0.62
0.26
B
0.91
0
1.53
D
0.62
1.53
0
0.36
0 volts
0.10 volts
0.26 volts
0.36 volts
0.55 volts
0.62 volts
0.65 volts
0.88 volts
0.98 volts
1.17 volts
1.27 volts
1.79 volts
1.89 volts

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, JJlover1892
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 15:30, alaf05160
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks. energy was destroyed inside the blocks. energy was absorbed into the blocks from outside the system. energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 17:20, banna01man
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2

Chemistry, 22.06.2019 19:00, ecolifesfsu1263
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1
You know the right answer?
Analyze and solve this partially completed galvanic cell puzzle. There are 4 electrodes each identif...
Questions in other subjects:


Mathematics, 15.07.2020 02:01

Mathematics, 15.07.2020 02:01

Mathematics, 15.07.2020 02:01

Mathematics, 15.07.2020 02:01




Mathematics, 15.07.2020 02:01

Mathematics, 15.07.2020 02:01