Chemistry, 21.04.2020 15:51 judyandaub1
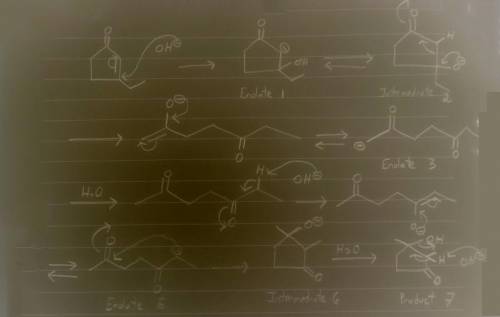
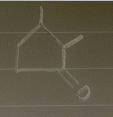
Aldol condensation of 2,5-heptanedione yields a mixture of two enone products in a 9:1 ratio. Treatment of the minor product with aqueous NaOH converts it into the major product; the interconversion proceeds as follows: Hydroxide ion adds to the double bond, forming enolate ion 1; Proton transfer occurs, yielding tetrahedral intermediate 2; Ring opening occurs, yielding enolate ion 3; Protonation of enolate ion 3 occurs, yielding 2,5-heptanedione; Deprotonation at C-6 occurs, yielding enolate ion 5; Enolate ion 5 attacks C-2, yielding tetrahedral intermediate 6; Protonation occurs to yield aldol addition product 7; Dehydration yields the more stable product.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, monnicawilliam
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 21.06.2019 20:50, britotellerialuis
Evaluate this exponential expression,8. (2 + 3)2 – 42
Answers: 3

Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 06:00, kylieweeks052704
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2
You know the right answer?
Aldol condensation of 2,5-heptanedione yields a mixture of two enone products in a 9:1 ratio. Treatm...
Questions in other subjects:

Mathematics, 05.12.2019 12:31

Chemistry, 05.12.2019 12:31

English, 05.12.2019 12:31

Mathematics, 05.12.2019 12:31


Mathematics, 05.12.2019 12:31


Social Studies, 05.12.2019 12:31