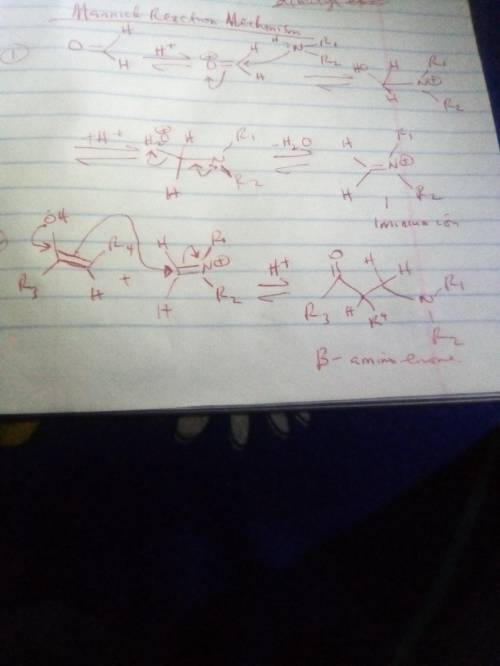
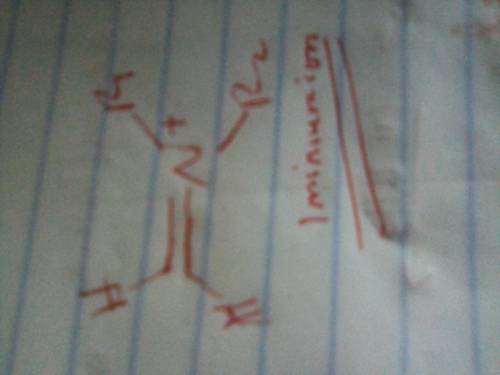
The Mannich reaction is one of the few three-component reactions in organic chemistry. In this reaction, a ketone, an aldehyde and an amine react together under acid catalyzed conditions to form the final product. The mechanism involves the following steps: 1. Following initial protonation of the carbonyl oxygen, nucleophilic attack by the amine forms a protonated carbinolamine 1; 2. Proton transfer and elimination of water forms iminium ion 2; 3. The enol form of the ketone attacks the iminium ion to form adduct 3; 4. Deprotonation of adduct 3 leads to the final product. Write out the mechanism on a separate sheet of paper and then draw the structure of iminium ion 2.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, lasagnafoe
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2



Chemistry, 23.06.2019 00:00, chloe8979
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1
You know the right answer?
The Mannich reaction is one of the few three-component reactions in organic chemistry. In this react...
Questions in other subjects:

Mathematics, 08.04.2020 18:31



History, 08.04.2020 18:32

History, 08.04.2020 18:32



Chemistry, 08.04.2020 18:32