
Chemistry, 21.04.2020 15:33 alexus6339
Consider a 125 mL buffer solution at 25°C that contains 0.500 mol of hypochlorous acid (HOCl) and 0.500 mol of sodium hypochlorite (NaOCl). What will be the pH of this buffer solution after adding 0.341 mol of HCl? The Ka of hypochlorous acid is 2.9 x 10-8 . Assume the change in volume of the buffer solution is negligible

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:30, SavageKidKobe
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1


Chemistry, 23.06.2019 04:20, tatta95
Which activity describes an application of topographic maps? check all that apply. recreation, such as camping and hiking engineering, such as the construction of roads and buildings science, such as mapping stars in the sky business, such as analyzing population centers science, such as analyzing surface features
Answers: 1
You know the right answer?
Consider a 125 mL buffer solution at 25°C that contains 0.500 mol of hypochlorous acid (HOCl) and 0....
Questions in other subjects:

Mathematics, 08.02.2021 23:20

Mathematics, 08.02.2021 23:20

English, 08.02.2021 23:20







![\frac{[NaOCl]}{[HOCl]}](/tpl/images/0614/6948/a0bd5.png)
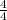 pH = 7.54
pH = 7.54![\frac{[NaOCl-HCl]}{[HOCl+HCl]}](/tpl/images/0614/6948/7bc1b.png) pH = 7.54 + log
pH = 7.54 + log 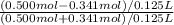 pH = 6.82
pH = 6.82

