Chemistry, 21.04.2020 00:24 vandonquisenberry
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 215. kJ during a certain chemical reaction, which is carried out at a constant pressure. Furthermore, by carefully monitoring the volume change it is determined that -155. kJ of work is done on the mixture during the reaction. Calculate the change in energy of the gas mixture during the reaction. Round your answer to 2 significant digits. Is the reaction exothermic or endothermic?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 01:30, Sonicawesomeness
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2

Chemistry, 23.06.2019 05:00, andrwisawesome0
Match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) amount of product predicted to be produced by the given reactants theoretical yield c) reactant that can produce more of the product
Answers: 3

Chemistry, 23.06.2019 05:00, lraesingleton
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 215. kJ during a...
Questions in other subjects:


World Languages, 16.03.2020 06:55

Chemistry, 16.03.2020 06:55

Mathematics, 16.03.2020 06:55

Biology, 16.03.2020 06:55

Mathematics, 16.03.2020 06:55

English, 16.03.2020 06:56


Biology, 16.03.2020 07:01

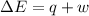
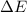 =Change in internal energy
=Change in internal energy