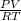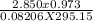Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, vapelordcarl69
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3



Chemistry, 22.06.2019 21:30, starl0rd211
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2
You know the right answer?
A sample of dry gas weighing 2.1025 g is found to occupy 2.850 L at 22.0oC and 740.0 mm Hg. How many...
Questions in other subjects:

Chemistry, 17.03.2020 23:26




Advanced Placement (AP), 17.03.2020 23:26

Physics, 17.03.2020 23:26


Mathematics, 17.03.2020 23:26

English, 17.03.2020 23:26