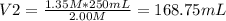You need to prepare 250.0 mL of a 1.35 M HCl solution from a 2.00 M HCl stock solution.
...

Chemistry, 16.04.2020 04:56 Bjehnsen3720
You need to prepare 250.0 mL of a 1.35 M HCl solution from a 2.00 M HCl stock solution.
a. Which glassware should you use to make the solution?
A. beaker
B. Erlenmeyer flask
C. volumetric flask
b. How should the correct amount of stock be obtained?
A. Measure out x g on a balance
B. Measure out x mL using a volumetric pipet
C. Measure out x mL using a graduated cylinder
c. Based on your answer above, what is the value of x?
d. How should the solution be mixed together?
A. Fill the container to the 250 mL mark then add the correct amount of stock solution.
B. Add the correct amount of stock solution then fill to the 250 mL mark with water.
C. Fill the container partially with water, add the correct amount of stock solution, then fill to the 250 mL mark with water.
D. None of these is the correct way to mix the stock solution.

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 23.06.2019 07:00, jpaintballer1
Agas has an empirical formula ch4. 0.16g of the gas occupies a volume of 240cm^3 what is the molecular formula of the me anyone who !
Answers: 1
You know the right answer?
Questions in other subjects:


Social Studies, 03.09.2019 04:20




Social Studies, 03.09.2019 04:20


Biology, 03.09.2019 04:20


Computers and Technology, 03.09.2019 04:20