Chemistry, 16.04.2020 01:23 cschellfamily
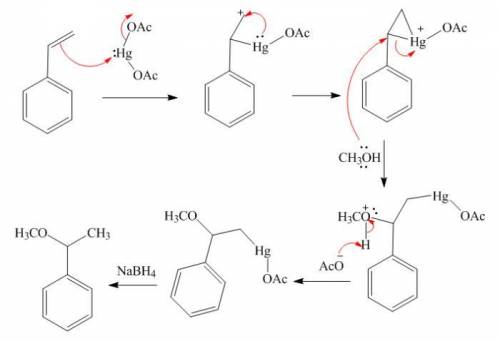
Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)2 (5.30 g, 0.016 mol) in methanol (100 mL) at room temp. and stirred for 24 h. Sodium hydroxide (3.0 M, 16 mL) was added, followed by NaBH4 (0.32 g, 0.008 mol) in NaOH (3.0 M, 16 mL) at 0 °C. The precipitated Hg was removed by filtration. The product was isolated by diethyl ether extraction. After drying over Na2SO4, solvent was removed and distillation gave the product. (Adapted from: Senda, Y.; Kanto, H.; Itoh, H. J. Chem. Soc., Perkin Trans. 2 1997, 1143-1146.)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, bernicewhite156
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2


Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1
You know the right answer?
Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)2 (5.30 g, 0.016 mo...
Questions in other subjects:

History, 14.12.2020 07:20

Social Studies, 14.12.2020 07:20



Mathematics, 14.12.2020 07:20



Arts, 14.12.2020 07:20

Chemistry, 14.12.2020 07:20