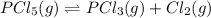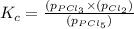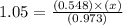Chemistry, 15.04.2020 21:03 soleydyperez
The equilibrium constant (Kp) for the decomposition of phosphorus pentachloride (PCl5) to phosphorus trichloride (PCl3) and molecular chlorine (Cl2) is found to be 1.05 at 250oC. If the equilibrium partial pressure of PCl5 and PCl3 are 0.973 and 0.548 atm, respectively, what is the equilibrium partial pressure of Cl2 at 250 oC?PCl5 (g) ↔ PCl3 (g) + Cl2 (g)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:20, montanolumpuy
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d. lytic
Answers: 1
You know the right answer?
The equilibrium constant (Kp) for the decomposition of phosphorus pentachloride (PCl5) to phosphorus...
Questions in other subjects:



Mathematics, 25.05.2021 16:00

Biology, 25.05.2021 16:00

Mathematics, 25.05.2021 16:00



Physics, 25.05.2021 16:00

Health, 25.05.2021 16:00

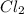 is 1.86 atm
is 1.86 atm