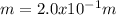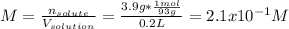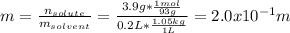Chemistry, 15.04.2020 18:54 samueldfhung
A student dissolves 3.9g of aniline (C6H5NH2) in 200.mL of a solvent with a density of 1.05 g/mL . The student notices that the volume of the solvent does not change when the aniline dissolves in it.
Calculate the molarity and molality of the student's solution. Be sure each of your answer entries has the correct number of significant digits.
How do I enter a number in scientific notation?
a. molarity =
b. molality =

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, kakesheco4210
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 04:00, fantasticratz2
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 23:00, soccerplayer17
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2
You know the right answer?
A student dissolves 3.9g of aniline (C6H5NH2) in 200.mL of a solvent with a density of 1.05 g/mL . T...
Questions in other subjects:

English, 21.08.2020 14:01

Geography, 21.08.2020 14:01

Mathematics, 21.08.2020 14:01

Computers and Technology, 21.08.2020 14:01


Social Studies, 21.08.2020 14:01


Mathematics, 21.08.2020 14:01