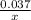Chemistry, 15.04.2020 04:48 anitaabbey27
Linolenic acid (C18H30O2) can be hydrogenated to stearic acid by reacting it with hydrogen gas according to the equation: C18H30O2 + 3H2 --->C18H36O2 What volume of hydrogen gas, measured at STP, is required to react with 10.5 g of linolenic acid in this reaction?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, isalih7256
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1

Chemistry, 22.06.2019 07:30, eburnhisel2023
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 22.06.2019 17:00, calmicaela12s
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 23.06.2019 05:00, pmbeachy3102
If 15 drops of ethanol from a medicine dropper weigh 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? density of ethanol is ethanol is 0.80g/ml.
Answers: 2
You know the right answer?
Linolenic acid (C18H30O2) can be hydrogenated to stearic acid by reacting it with hydrogen gas accor...
Questions in other subjects:

Mathematics, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00


Mathematics, 19.11.2020 19:00

Mathematics, 19.11.2020 19:00





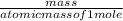
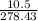
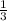 =
=