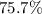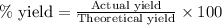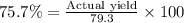Chemistry, 15.04.2020 03:45 danieldfuenteg732
If 36.9 mL of B2H6 reacted with excess oxygen gas, determine the actual yield of B2O3 if the percent yield of B2O3 was 75.7%. (The density of B2H6 is 1.131 g/mL. The molar mass of B2H6 is 27.668 g/mol and the molar mass of B2O3 is 69.62 g/mol.)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, Usman458
The twister and runaway train are two coasters at the same amusement park. both coasters start at the same height. the coaster for the twister is twice the mass of the coaster for the runaway train. which roller coaster has greater gravitational potential energy at the start of the ride?
Answers: 1

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 22:00, jespinozagarcia805
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a. rectant b. product c. supernate
Answers: 3
You know the right answer?
If 36.9 mL of B2H6 reacted with excess oxygen gas, determine the actual yield of B2O3 if the percent...
Questions in other subjects:



Mathematics, 05.05.2020 18:04



Biology, 05.05.2020 18:04

Mathematics, 05.05.2020 18:04



Arts, 05.05.2020 18:04

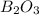 is 60.0 g
is 60.0 g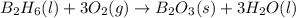
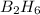 =
=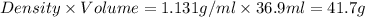
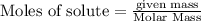
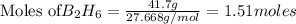
 moles of
moles of