Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide:
...

Chemistry, 15.04.2020 03:36 Crtive5515
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide:
CaO(s)+H2O(l)→Ca(OH)2(s)
In a particular experiment, a 1.00-g sample of CaO is reacted with excess water and 0.82 g of Ca(OH)2 is recovered. What is the percent yield in this experiment?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:10, tishfaco5000
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 08:30, vanessadaniellet21
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

You know the right answer?
Questions in other subjects:

History, 30.09.2019 07:30



Mathematics, 30.09.2019 07:30

English, 30.09.2019 07:30

Mathematics, 30.09.2019 07:30


Biology, 30.09.2019 07:30


Mathematics, 30.09.2019 07:30

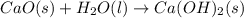
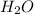 is the excess reagent,
is the excess reagent, 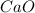 acts as the limiting reagent and it limits the formation of product.
acts as the limiting reagent and it limits the formation of product.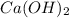
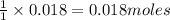 of
of