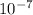Chemistry, 15.04.2020 03:41 sammysosa121832
The energy needed to ionize an atom of si when it is in the most stable is 786.4 kJ mol^-1 however if an atom of Si is in certain low lying excited state only 310.8 is needed to ionize.
what is the wavelength of he radiation emitted when an atom of si undergoes a transition from this excited state to the ground state?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, petriajack8375
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3



Chemistry, 23.06.2019 02:30, elyzarobertson
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
The energy needed to ionize an atom of si when it is in the most stable is 786.4 kJ mol^-1 however i...
Questions in other subjects:

Mathematics, 27.10.2019 12:43


Biology, 27.10.2019 12:43



Mathematics, 27.10.2019 12:43

Mathematics, 27.10.2019 12:43

Mathematics, 27.10.2019 12:43

Mathematics, 27.10.2019 12:43

Social Studies, 27.10.2019 12:43

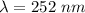

 ----- (1)
----- (1)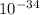 J s
J s
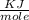

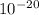 J
J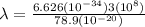
 ×
×