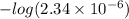Chemistry, 15.04.2020 00:52 calebcoolbeans6691
A chemist titrates 160.0 mL of a 0.3065 M cyanic acid (HCNO) solution with 0.4994 M NaOH solution at 25 °C. Calculate the pH at equivalence. The pK of cyanic acid is 3.46 Round your answer to 2 decimal places.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:50, sgslayerkingminecraf
Which of the following statements about acidic water is true? a. acid has no effect on the h, o molecules. b. the solution contains a larger number of oh ions than h, o ions. c. the solution contains a larger number of h, o ions than qh ions. d. the solution contains an equal number of h, o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 11:30, elijah1090
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 19:00, hmontalvo22
How many moles are contained in 5.6 l of h2 at stp
Answers: 3

You know the right answer?
A chemist titrates 160.0 mL of a 0.3065 M cyanic acid (HCNO) solution with 0.4994 M NaOH solution at...
Questions in other subjects:




Social Studies, 31.03.2020 05:00



History, 31.03.2020 05:00


History, 31.03.2020 05:00

Arts, 31.03.2020 05:00

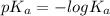
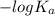
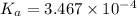
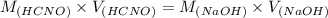
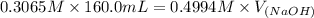
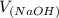 = 98.1978 mL
= 98.1978 mL
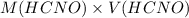
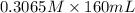
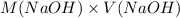
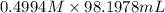
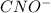 and
and 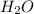 .
.
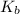 of
of 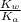
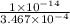

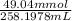
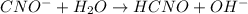
![K_{b} = \frac{[HCNO][OH^{-}]}{[CNO^{-}]}](/tpl/images/0600/3810/7fa50.png)
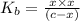
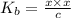
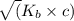
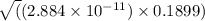
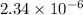
![[OH^{-}] = x = 2.34 \times 10^{-6}](/tpl/images/0600/3810/bb369.png) M
M
![-log [OH^{-}]](/tpl/images/0600/3810/337a4.png)