
Chemistry, 14.04.2020 21:42 jybuccaneers2022
Diiodine pentoxide (I2O5) is used in respirators to remove carbon monoxide (CO) from air according to the chemical equation: I2O5 (s) + 5 CO (g) --> I2 (s) + 5 CO2 (g) What mass of carbon monoxide could be removed from air by a respirator that contains 15.9 g of diiodine pentoxide? The molar mass of I2O5 is 333.8 g / mol.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kingteron5870
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2
You know the right answer?
Diiodine pentoxide (I2O5) is used in respirators to remove carbon monoxide (CO) from air according t...
Questions in other subjects:



English, 05.12.2021 19:50


History, 05.12.2021 19:50

Chemistry, 05.12.2021 20:00

English, 05.12.2021 20:00


History, 05.12.2021 20:00

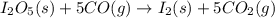
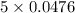 of CO from air
of CO from air

