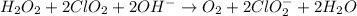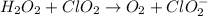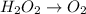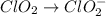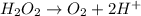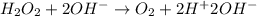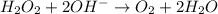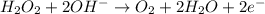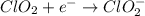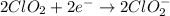Chemistry, 14.04.2020 20:14 cadereymer24
Given the partial equation: H2O2 + ClO2 → O2 + ClO2−, balance the reaction in basic solution using the half-reaction method and fill in the coefficients. The missing blanks represent H2O, H+, or OH-, as required to balance the reaction. Enter the coefficients as integers, using the lowest whole numbers. If the coefficient for something is "1", make sure to type that in and not leave it blank. Enter only the coefficients. H2O2 + ClO2 + → O2 + ClO2− +

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:20, nyceastcoast
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1
You know the right answer?
Given the partial equation: H2O2 + ClO2 → O2 + ClO2−, balance the reaction in basic solution using t...
Questions in other subjects:

English, 02.10.2019 09:50


World Languages, 02.10.2019 09:50

Mathematics, 02.10.2019 09:50

English, 02.10.2019 09:50



Mathematics, 02.10.2019 09:50