Chemistry, 14.04.2020 17:28 marifermolina
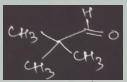
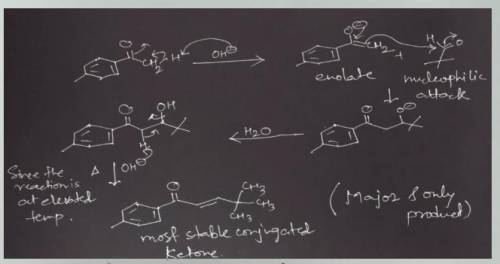
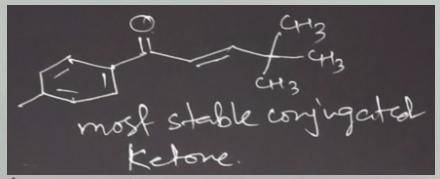
The aqueous hydroxide-promoted reaction of the compound with 2,2-dimethylpropanal (pivalaldehyde) at elevated temperature gives only one product. Write the mechanism of the reaction and the structure of the final product. Would you mix hydroxide with the ketone and then add the aldehyde, or would you mix hydroxide with the aldehyde and then add the ketone?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:10, cheesedoodle
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3
You know the right answer?
The aqueous hydroxide-promoted reaction of the compound with 2,2-dimethylpropanal (pivalaldehyde) at...
Questions in other subjects:



World Languages, 19.08.2021 21:00




Social Studies, 19.08.2021 21:00

Mathematics, 19.08.2021 21:00

History, 19.08.2021 21:00

Mathematics, 19.08.2021 21:00

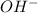 is first made to react with the ketone group
is first made to react with the ketone group