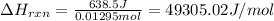Chemistry, 31.01.2020 02:45 rileyeddins1010
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride according to the reaction: agno3(aq)+hcl(aq)→agcl(s)+hno3(aq). when you combine 70.0ml of 0.185m agno3 with 70.0ml of 0.185m hcl in a coffee-cup calorimeter, the temperature changes from 23.16∘c to 24.25∘c.. calculate δhrxn for the reaction as written. use 1.00 g/ml as the density of the solution and c=4.18j/g⋅∘c as the specific heat capacity.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, angeljohnson2081
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1



Chemistry, 22.06.2019 18:30, kate3887
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride accordin...
Questions in other subjects:





History, 09.07.2019 05:00

Biology, 09.07.2019 05:00

Mathematics, 09.07.2019 05:00

Mathematics, 09.07.2019 05:00


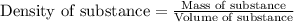
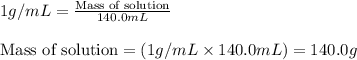
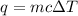
 = change in temperature =
= change in temperature = ![[24.25-23.16]^oC=1.09^oC](/tpl/images/0488/1311/2253a.png)
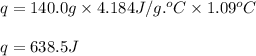
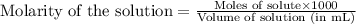
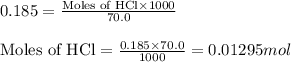
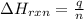
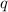 = amount of heat absorbed = 638.5 J
= amount of heat absorbed = 638.5 J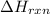 = enthalpy change of the reaction
= enthalpy change of the reaction