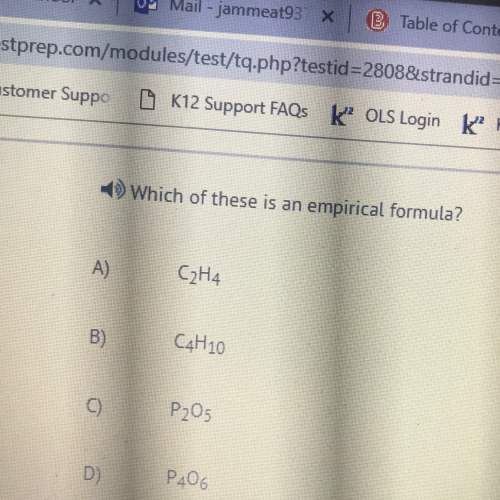
Which of the following halogens would have the highest melting point?
a. f2
b. cl2
c....

Chemistry, 11.04.2020 10:45 makrosebud7821
Which of the following halogens would have the highest melting point?
a. f2
b. cl2
c. br2
d. i2

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 20:30, jaydenbrock
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Questions in other subjects:

Biology, 01.09.2019 09:00


History, 01.09.2019 09:00

World Languages, 01.09.2019 09:00



History, 01.09.2019 09:00

Computers and Technology, 01.09.2019 09:00

History, 01.09.2019 09:00