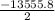Chemistry, 11.04.2020 01:50 smileyjesse6073
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for decane is —300.9 kJ/mol using the balanced chemical equation and Appendix Table B–14.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:00, jessicannoh5965
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 22.06.2019 19:30, youngdelvin123
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 22.06.2019 21:00, melissalopez12
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
You know the right answer?
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for dec...
Questions in other subjects:

Social Studies, 29.01.2020 23:53

History, 29.01.2020 23:53

History, 29.01.2020 23:53



Mathematics, 29.01.2020 23:53



English, 29.01.2020 23:53

Biology, 29.01.2020 23:53

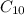
 + 31
+ 31  ⇒ 20 C
⇒ 20 C O
O