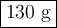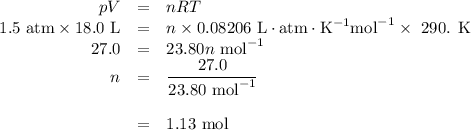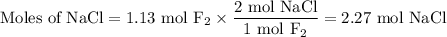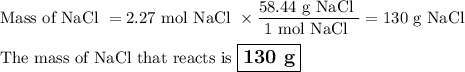Chemistry, 11.04.2020 00:53 kuehnkeegan
I need this answered ASAP!
Part 1. A chemist reacted 18.0 liters of F2 gas with NaCl in the laboratory to form Cl2 gas and NaF. Use the ideal gas law equation to determine the mass of NaCl that reacted with F2 at 290. K and 1.5 atm. F2 + 2NaCl → Cl2 + 2NaF
Part 2. Explain how you would determine the mass of sodium chloride that can react with the same volume of fluorine gas at STP.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:10, danielahchf
How is 0.00235 expressed in proper scientific notation? a. 2.35 × 10-3 b. 0.235 × 10-2 c. 2.35 d. 2.35 × 103
Answers: 1



Chemistry, 22.06.2019 17:40, Snowball080717
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
I need this answered ASAP!
Part 1. A chemist reacted 18.0 liters of F2 gas with NaCl in the la...
Part 1. A chemist reacted 18.0 liters of F2 gas with NaCl in the la...
Questions in other subjects:

Mathematics, 27.06.2019 09:00


Biology, 27.06.2019 09:00


Mathematics, 27.06.2019 09:00



Biology, 27.06.2019 09:00

Mathematics, 27.06.2019 09:00

Mathematics, 27.06.2019 09:00