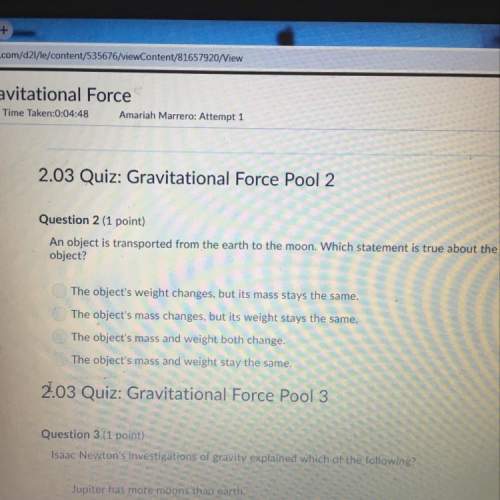
B
Acid rain is a dilute solution of acids that dissolve
the calcium carbonate in limesto...

Chemistry, 09.04.2020 06:28 Bra1nPowers
B
Acid rain is a dilute solution of acids that dissolve
the calcium carbonate in limestone statues.
Concentrated acids can dissolve a large piece of
limestone in a few days. Statue breakdown due to
acid rain can take decades, but statues with
intricate carvings break down more quickly.
Explain these observations in terms of reaction
rates.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:00, kajjumiaialome
Note the ph and poh values labeled with letters on the ph scale below. based on log rules and the way ph is calculated, what is the difference in [oh– ] concentration between point a and point b. a) 10^1 b) 10^5 c) 10^6 d) 10^7
Answers: 1



You know the right answer?
Questions in other subjects:


Geography, 25.06.2019 19:20

Mathematics, 25.06.2019 19:20

Mathematics, 25.06.2019 19:20

Computers and Technology, 25.06.2019 19:20


Mathematics, 25.06.2019 19:20