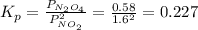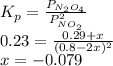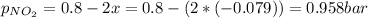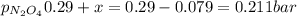N2O4 decomposes into NO2. At a certain temperature, the equilibrium pressures of NO2 and N2O4 are 1.6 bar and 0.58 bar, respectively. If the volume of the container is doubled at constant temperature, what would be the partial pressures of the gases when equilibrium is re-established

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, pettygirl13
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3


Chemistry, 22.06.2019 12:30, meghan2529
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 22:30, kristen17diaz
How many valence electrons are in atom of radon?
Answers: 1
You know the right answer?
N2O4 decomposes into NO2. At a certain temperature, the equilibrium pressures of NO2 and N2O4 are 1....
Questions in other subjects:


Mathematics, 11.11.2020 07:10


Mathematics, 11.11.2020 07:10

English, 11.11.2020 07:10

Biology, 11.11.2020 07:10


Mathematics, 11.11.2020 07:10