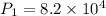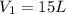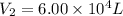Chemistry, 08.04.2020 17:17 clydeben4543
A 15.0 L tank of gas is contained at a high pressure of 8.20*10^4. The tank is opened and the gas expands into an empty chamber with a volume of 6.00*10^4 L. Calculate the new pressure of the gas.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, peternice2956
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1


Chemistry, 22.06.2019 14:50, jonmorton159
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3
You know the right answer?
A 15.0 L tank of gas is contained at a high pressure of 8.20*10^4. The tank is opened and the gas ex...
Questions in other subjects:

Mathematics, 27.08.2019 06:00

Geography, 27.08.2019 06:00

Mathematics, 27.08.2019 06:00

Health, 27.08.2019 06:00

Mathematics, 27.08.2019 06:00


Mathematics, 27.08.2019 06:00




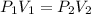 ..........1
..........1